Cutting Batch Release from 14 Days to 3: A Case Study in Multi-Agent AI for Pharmaceutical Manufacturing
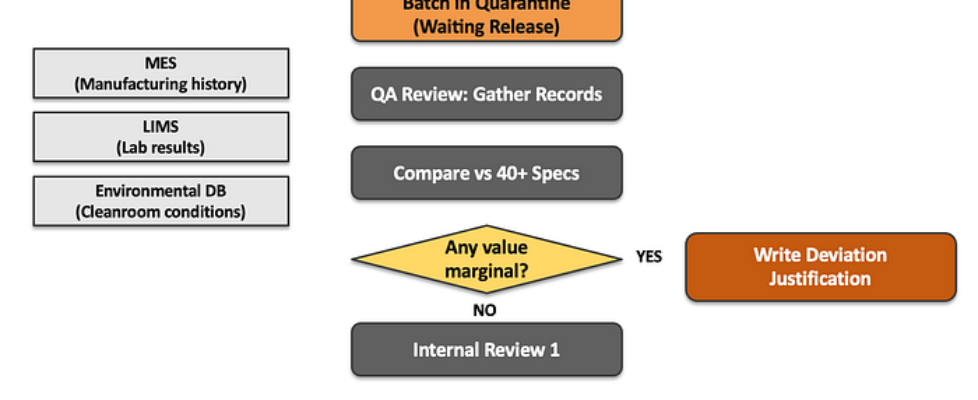
Author(s): Saif Ali Kheraj Originally published on Towards AI. How agentic AI architectures can compress decision cycles in regulated manufacturing with a practical implementation blueprint using CrewAI. Every contract pharmaceutical manufacturer faces the same operational bottleneck. After a medicine batch completes production and passes all laboratory tests, it moves into a quarantine status not because there is any defect in the drug, but because regulatory review has not yet been completed. Figure 1: Process Management by AuthorThis case study details how XYZ Pharma implemented a multi-agent AI system, reducing the quarantine period for drug batches from 14 days to 3 days while maintaining compliance with regulatory standards. It highlights the traditional processes that lead to delays, such as the need for meticulous review of production and lab data by Quality Assurance (QA) teams, and explores the innovative use of AI technologies to streamline decision-making and enhance operational efficiency in the pharmaceutical manufacturing sector. Read the full blog for free on Medium. Join thousands of data leaders on the AI newsletter. Join over 80,000 subscribers and keep up to date with the latest developments in AI. From research to projects and ideas. If you are building an AI startup, an AI-related product, or a service, we invite you to consider becoming a sponsor. Published via Towards AI